21 HPV GenoArray Diagnostic Kit
21 HPV GenoArray Diagnostic Kit
Ref: HBGA-21
![]()
The 21 HPV GenoArray Diagnostic Kit is a qualitative PCR based in vitro diagnostic test for identification of 21 HPV genotyping in cervical specimens. The assay is optimized to detect 15 high-risk HPV and 6 low-risk HPV.
By using polymerase chain reaction (PCR) to amplify extracted HPV DNA from cervical samples, amplified DNA amplicons are hybridized with specific HPV probes located inside the ‘HybriMem’ under our US patented “flow-through hybridization” technology, results are obtained by colorimetric result using enzyme immunoassay method.
Description
Features
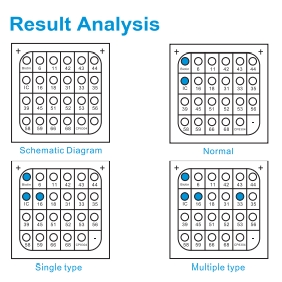
- Detection of 21 HPV genotypes in one single reaction
- CE-IVD marked and CFDA registered
- WHO HPV LabNet Proficiency Test validated (2011, 2013, 2015 and 2017)
- The only HPV assay appointed by The CervicalCancer Prevention Program in China (Over three million tests performed in 2018)
- High clinical sensitivity and clinical specificity: >95% (compared to FDA approved kit)
- Promising results comparing to Roche’s Linear Array; with a better detection of HPV 52
- Include amplification control (IC) and hybridisation control (Biotin) for monitoring of the entire experimental process
HPV type covering
- 15 High-risk genotypes:HPV 16, 18, 31, 33, 35, 39, 45, 51, 52, 53, 56, 58, 59, 66, 68
- 6 Low-risk genotypes:HPV 6, 11, 42, 43, 44, 81
Specimens
- Cervical swab (pap smear)
- Liquid based cytology specimen (e.g. Thin prep, Sure path, Roche Female Swab Sample Kit, Hybribio Famale Sample Collection Kit)
- Urine sample
- Exfoliated skin cells using sandpaper around genital area (Male)
Benefits
- Rapid and accurate identification of multiple HPV types in a single reaction
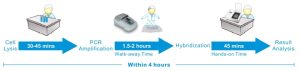
- Easy to operate with <1.5 hours hands-on time
- Triage and management of patients with ASCUS & LSIL
- Efficient and cost-effective